
Scientists had now established that the atom was not indivisible as Dalton had believed.īut the structure of the atom was otherwise not well understood. Further work with charge to mass ratios allowed him to determine the mass of the electron as well. By adjusting the electric field strength and making careful measurements and appropriate calculations, Millikan was able to determine the charge on individual drops ( Figure 2), and figure out the charge on individual electrons. Millikan created microscopic oil droplets, which could be electrically charged by friction, set to drop due to gravity, and then slowed or even reversed by an applied electric field lower in the apparatus. In 1909, more information about the electron was uncovered by American physicist Robert A. The term “electron” was coined in 1891 by Irish physicist George Stoney, from “ electric ion.”Ĭlick here to hear Thomson describe his discovery in his own voice. Furthermore, they are less massive than atoms and indistinguishable, regardless of the source material, so they must be fundamental, subatomic constituents of all atoms.Īlthough controversial at the time, Thomson’s idea was gradually accepted, and his cathode ray particle is what we now call an electron, a negatively charged, subatomic particle with a mass approximately two thousands that of a hydrogen atom. (credit a: modification of work by Nobel Foundation credit b: modification of work by Eugen Nesper credit c: modification of work by “Kurzon”/Wikimedia Commons)īased on his observations, Thomson proposed that the particles are attracted by positive (+) charges and repelled by negative (−) charges, so they must be negatively charged (like charges repel and unlike charges attract). Simultaneous deflections by applied electric and magnetic fields permitted Thomson to calculate the mass-to-charge ratio of the particles composing the cathode ray. (c) In the cathode ray, the beam (shown in yellow) comes from the cathode and is accelerated past the anode toward a fluorescent scale at the end of the tube. 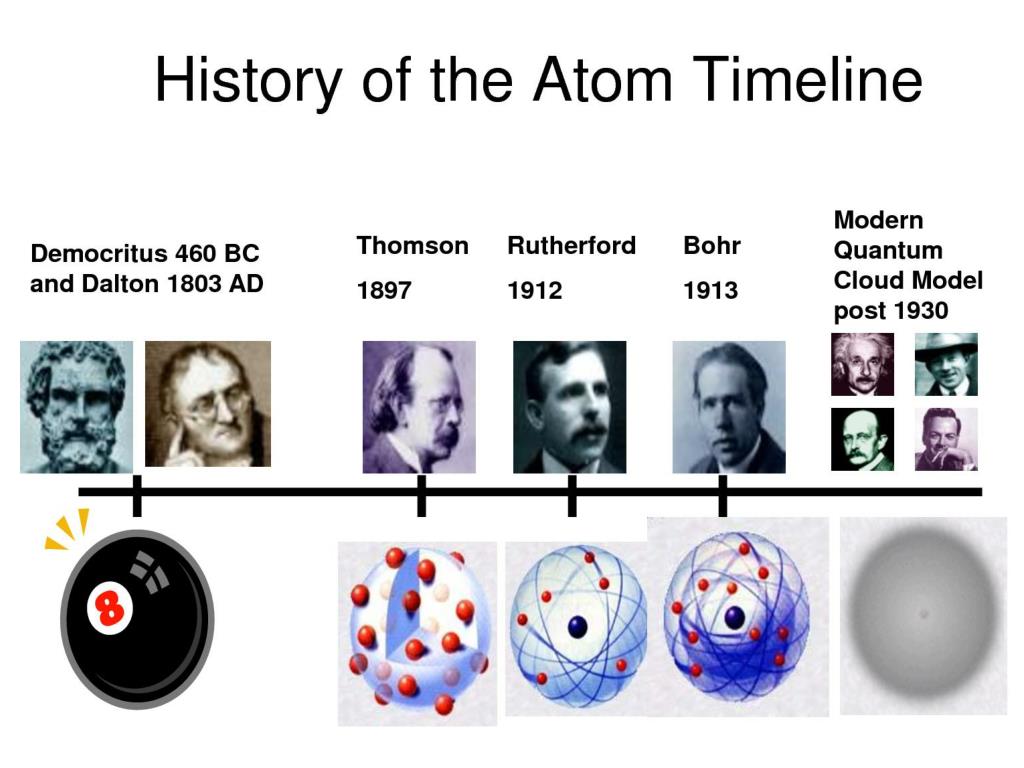
(b) This is an early cathode ray tube, invented in 1897 by Ferdinand Braun. Thomson produced a visible beam in a cathode ray tube. The results of these measurements indicated that these particles were much lighter than atoms ( Figure 1). In similar experiments, the ray was simultaneously deflected by an applied magnetic field, and measurements of the extent of deflection and the magnetic field strength allowed Thomson to calculate the charge-to-mass ratio of the cathode ray particles. This beam was deflected toward the positive charge and away from the negative charge, and was produced in the same way with identical properties when different metals were used for the electrodes. When high voltage was applied across the electrodes, a visible beam called a cathode ray appeared between them. This apparatus consisted of a sealed glass tube from which almost all the air had been removed the tube contained two metal electrodes. If matter were composed of atoms, what were atoms composed of? Were they the smallest particles, or was there something smaller? In the late 1800s, a number of scientists interested in questions like these investigated the electrical discharges that could be produced in low-pressure gases, with the most significant discovery made by English physicist J. Atomic Theory after the Nineteenth Century An important adjustment to Dalton’s Theory: atoms are divisible Much of this came from the results of several seminal experiments that revealed the details of the internal structure of atoms. In the two centuries since Dalton developed his ideas, scientists have made significant progress in furthering our understanding of atomic theory. Define isotopes and give examples for several elements.Describe the three subatomic particles that compose atoms.Outline milestones in the development of modern atomic theory. 
The evidence for atoms is so great that few doubt their existence.By the end of this section, you will be able to: \): Dalton's symbols from his text "A New System of Chemical Philosophy."
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
